Literatur
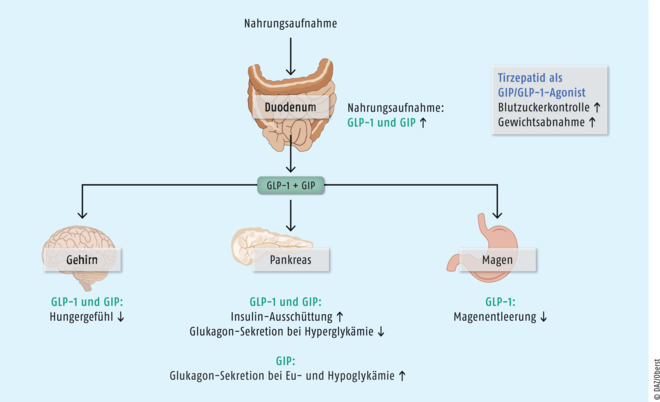
[1] Fachinformation zu Mounjaro®, Stand November 2023
[2] Rosenstock J, Wysham C, Frías JP et al. Efficacy and safety of a novel dual GIP and GLP-1 receptor agonist tirzepatide in patients with type 2 diabetes (SURPASS-1): a double-blind, randomised, phase 3 trial. Lancet 2021;398(10295):143-155, doi: 10.1016/S0140-6736(21)01324-6
[3] Frías JP, Davies MJ, Rosenstock J et al. Tirzepatide versus semaglutide once weekly in patients with type 2 diabetes. N Engl J Med 2021;385(6):503-515, doi: 10.1056/NEJMoa2107519
[4] Ludvik B, Giorgino F, Jódar E et al. Once-weekly tirzepatide versus once-daily insulin degludec as add-on to metformin with or without SGLT2 inhibitors in patients with type 2 diabetes (SURPASS-3): a randomised, open-label, parallel-group, phase 3 trial. Lancet 2021;398(10300):583-598, doi: 10.1016/S0140-6736(21)01443-4
[5] Del Prato S, Kahn SE, Pavo I et al. Tirzepatide versus insulin glargine in type 2 diabetes and increased cardiovascular risk (SURPASS-4): a randomised, open-label, parallel-group, multicentre, phase 3 trial. Lancet 2021;398(10313):1811-1824, doi: 10.1016/S0140-6736(21)02188-7
[6] Dahl D, Onishi Y, Norwood P et al. Effect of subcutaneous tirzepatide vs placebo added to titrated insulin glargine on glycemic control in patients with type 2 diabetes: the SURPASS-5 randomized clinical trial. JAMA 2022;327(6):534-545, doi:10.1001/jama.2022.0078
[7] EPAR summary for the public. Mounjaro® (Tirzepatid). Informationen der Europäischen Arzneimittel-Agentur, EMA/674324/2022
[8] Jastreboff AM, Aronne LJ, Ahmad NN et al. Tirzepatide Once Weekly for the Treatment of Obesity. N Engl J Med 2022;387:205-216, doi: 10.1056/NEJMoa2206038
[9] EPAR summary for the public. Saxenda® (Liraglutid). Informationen der Europäischen Arzneimittel-Agentur, EMA/291319/2023
[10] EPAR summary for the public. Wegovy® (Semaglutid). Informationen der Europäischen Arzneimittel-Agentur, EMA/162766/2023


























0 Kommentare
Das Kommentieren ist aktuell nicht möglich.